Ionic Bond — Formation & Compounds Expii
The structure is an ionic lattice, but it has a lot of covalent character. At atmospheric pressure, aluminum chloride sublimes at about 180°C. If the pressure is increased to just over 2 atmospheres, it melts instead at a temperature of 192°C. Both of these temperatures are far below the expected range for an ionic compound.

Ionic bond formation in AlCl3(alluminium trichloride) YouTube
When electrons are transferred and ions form, ionic bonds result. Ionic bonds are electrostatic forces of attraction, that is, the attractive forces experienced between objects of opposite electrical charge (in this case, cations and anions). When electrons are "shared" and molecules form, covalent bonds result.

Is AlCl3 ionic or covalent or both? What type of bond in Aluminium
Aluminum chloride is an inorganic compound. AlCl3 is an odorless and white/ pale yellow crystalline corrosive solid. It exists in a hydrated (AlCl3.6H2O) and an anhydrous form (AlCl3). In this article, we will understand the concepts of prediction of Lewis structure, geometry, hybridization, and polarity of a given compound. Contents show

Ionic Bond Definition, Types, Properties & Examples
Is Alcl3 Ionic or Covalent? View Solution Click here:point_up_2:to get an answer to your question :writing_hand:alcl3 is covalent where as alf3 is ionic why
Edexcel A Level Chemistry复习笔记1.4.6 Covalent DotandCross Diagrams翰林国际教育
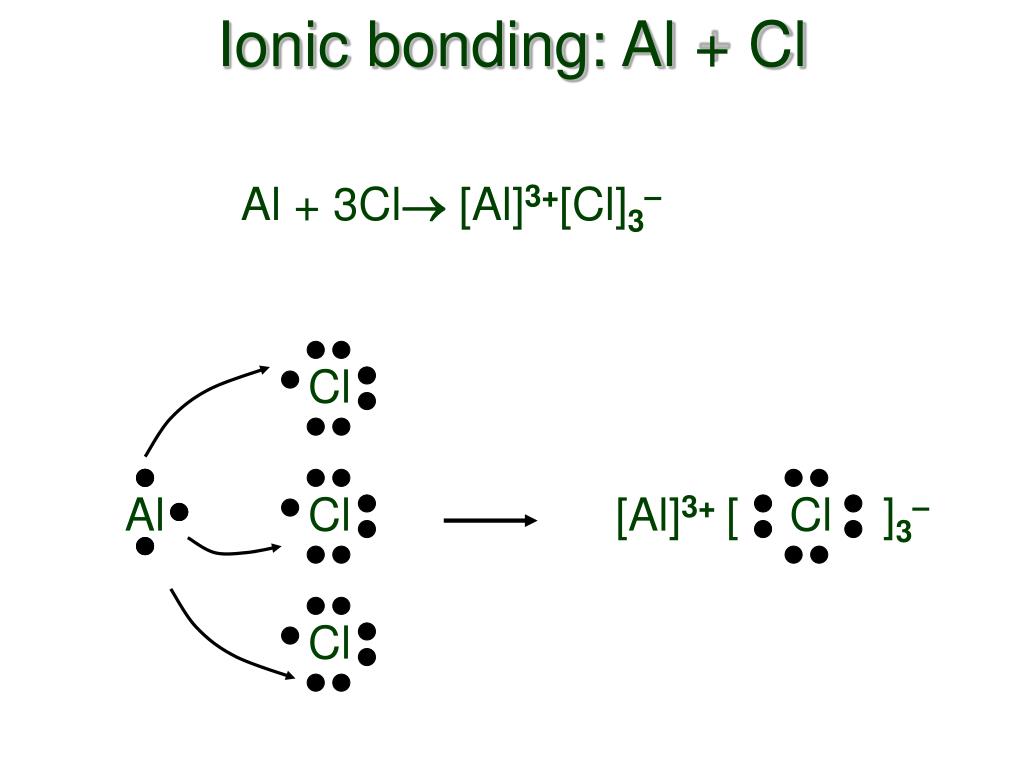
AlCl3 is an ionic compound because it contains an aluminum cation (Al3+) and a chloride anion (Cl-). The Al3+ ion has a charge of +3, while the Cl- ion has a charge of -1. This creates an unequal distribution of electrons between the two atoms, which forms an ionic bond. What Are Ionic And Covalent Bonds?
Anhydrous AlCl3 is covalent if the energy to ionize AlCl3 = 5215 kJ mol
Aluminium chloride, also known as aluminium trichloride, is an inorganic compound with the formula AlCl3. It forms a hexahydrate with the formula [Al (H2O)6]Cl3, containing six water molecules of hydration.

Leave a Comment Cancel Reply
Is Alcl3 Ionic or Covalent? Leave a Comment / Chemistry / By reta Chemical bonds are the forces that hold atoms together to form molecules. There are two main types of chemical bonds - ionic and covalent bonds.

Ionic vs Covalent Which is which and how to tell them apart
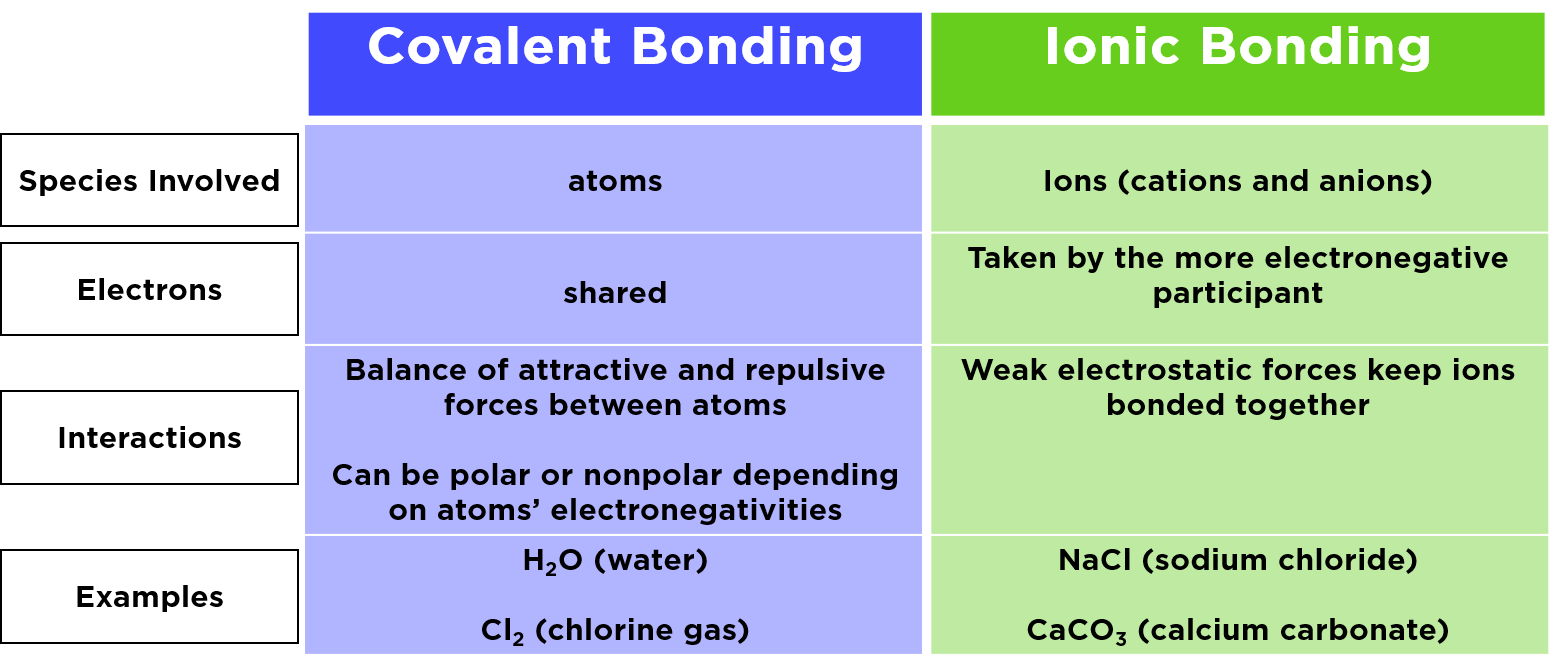
1 OH group is smaller than Cl and Al (OH)3 isn't molecular, unlike the chloride. - Mithoron Jun 28, 2022 at 19:39 In the pure state, |AlClX3 | A l C l X 3 dimerizes as a molecule AlX2ClX6 A l X 2 C l X 6. The structure is made of two squares having one edge in common (like an open book).
PPT Bonding, Electronegativity , & Bond Shape PowerPoint Presentation
Figure2.7.3 2.7. 3: Example of some compounds that have multiple oxidation states. Note mercury (1) is not a monoatomic cation, but is really a homonuclear diatomic ion of two mercury atoms bound to each other, both having lost one electron. So Hg 2 Cl 2 it the lowest whole number ratio of cation to anion. Example 2.

Which of the following is a covalent compound?
Ionic and molecular compounds are named using somewhat-different methods. Binary ionic compounds typically consist of a metal and a nonmetal. The name of the metal is written first, followed by the name of the nonmetal with its ending changed to - ide. For example, K 2 O is called potassium oxide.

Is AlCl3 Ionic or Covalent? Techiescientist
Compounds that contain ions are called ionic compounds. Ionic compounds generally form from metals and nonmetals. Compounds that do not contain ions, but instead consist of atoms bonded tightly together in molecules (uncharged groups of atoms that behave as a single unit), are called covalent compounds.

Anhydrous AlCl3 is covalent but hydrated AlCl3.6H2O is ionic because
Is AlCl3 Ionic or Covalent? Let us study the nature of an antiperspirant compound known as aluminum chloride. Aluminum chloride is also identified as aluminum trichloride. The formula of this compound is AlCl3 (H2O)n (n = 0 or 6). It has a molecular weight of 133.34. Anhydrous aluminum chloride is a white to gray powder that smells pungent.

How to Draw the Lewis Structure for AlCl3 Aluminum Chloride YouTube
Therefore AlF3 is ionic but AlCl3 is covalent. Electronegativity difference:- If Electronegativity difference between bounding atoms is less than 1.7 then bond has more covalent charactor. And Electronegativity difference is 1.55 between Al and Cl. Therefore Al——Cl bond is covalent. Suggest Corrections.
KWOK The Chem Teacher Chemical Bonding Dative Covalent and Expansion
1 Answer Sorted by: 6 The distinction between ionic and covalent compounds is arbitrary and many compounds fall somewhere in the middle. This is explained in a previous question. In the case of AlClX3 A l C l X 3 we have a metal 'cation' and a non-metal 'anion' so in a simplistic view we would expect this to be an ionic compound.

What is the electron dot structure of alcl3? Brainly.in
AlCl3 is not ionic, but a covalent compound. Because aluminum and three chlorine atoms are linked through the sharing of electrons with each other, although, aluminum (Al) is a metal and chlorine (Cl) is a nonmetal, still, there does not occur a complete transfer of electrons between the metal and the non-metal.

Difference Between Covalent and Ionic Bonds Gcse Chemistry Revision
Why does AlCl3 form a dimer? AlCl3 is borderline for being ionic/covalent. When solid it is usually ionic. In vapor it is covalent. In this state 3 electrons from Al are shared with 3 Cl electrons.